Build an atom5/10/2023 If students gather data quickly, I let them try the game, which presents them with numbers of particles and asks them to identify the element. I then let them experiment for about 15 minutes and discuss what they are observing. I remind them to flip their help cards to red if they get stuck. I show them how when they reset they need to re-open the Symbol, Mass, and Charge tabs before beginning. I instruct them to then try different combinations of particles in the space provided, resetting each time. I then show them how to reset the simulation. We record each observation in the correct column. I add 3 protons to the atom and ask the students what changes occur. Once all students are logged in and have a sheet, I use the projector to demonstrate the simulation ( HTML5 version) for them and we all do the first row of the worksheet together as seen in this screencapture. While students are logging into the computers, I distribute the Build an Atom Simulation worksheet and help cards. Our system often takes up to three minutes to log in, so this provides a transitional time. I ask students to go back with their partners and log into the computers. I then tell the class today we will be using the computers to build upon and enhance our understanding of the atom's structure. For instance, " How do you know there's a nucleus?" or " Why did you draw the electrons as minus signs?" I ask explicit questions to link their models with our experiments with charges, Thomson's cathode ray experiment, and Rutherford's Gold foil experiment. I select a few drawings to share via the document camera and we discuss the strengths of each model, and what evidence we've learned for each piece so far. Some students may represent both protons and neutrons from our vocabulary work, and most advanced students will have equal numbers of all particles. In previous lessons (linked here) we have "discovered" the positive nucleus and the presence of negative electrons already, so I am expecting students' drawings to represent a nucleus with electrons orbiting it. Once attendance is done, I circulate the room inspecting their drawings. When the bell rings, I ask students to find a blank page in their binders and complete the activity while I take attendance. Don't have the students exit the computers before the debrief so they are ready to go, and return to the computers after the debrief to play a couple round of the game for five minutes, and give an extra three minutes to the exit ticket.Īs students enter, the direction "Draw an atom on a piece of paper" is on the board. If you had extra time, I would recommend spending it on the simulation game with students. 
My district does department meetings each Wednesday morning, and our classes are shortened as a result. From their data, they will use Science and Engineering Practice 6 and construct an explanation from their simulation results - both individually and together in small groups. We also will be tapping into Science and Engineering Practice 4, Analyzing and interpreting data, as students will decide which particles to add in each trial, and record what is changing to determine the properties of each subatomic particle. However, this particular simulation is also available via HTML5, and runs on tablets, netbooks and smartphone browsers. It does require Java, so it may not be accessible in all locations. 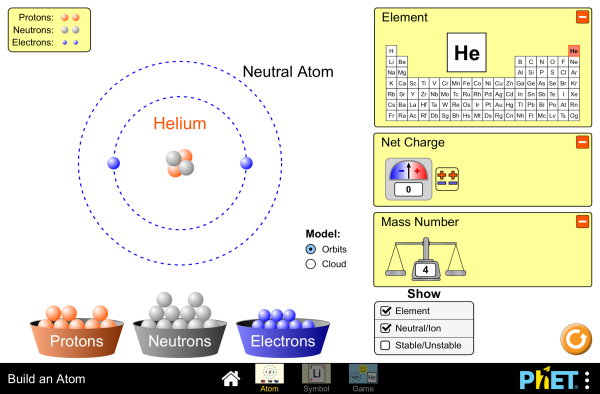
This is a free online simulation it that I use a lot throughout the year to provide clear visuals for students. Our model of choice is a computer model provided by the PhET project at the University of Colorado Boulder. Our emphasis today is on Science and Engineering Practice 2, Using models. Students will engage this after learning about the discoveries of the electron and the nucleus, but likely with an incomplete visual model of the atom. This is the first NGSS Performance Expectation that asks students to use the periodic table, and this activity begins to introduce them to the format of individual cells of the periodic table, and how protons are related to atomic number and identity of elements. This lesson is the first to begin to truly address HS PS1-1: Using the periodic table as a model to predict properties of elements based on the patterns of electrons in the outermost energy level of atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
